Free Chlorine vs Total Chlorine: Essential Knowledge for Water System Operators
Chlorine is an indispensable chemical in water treatment processes worldwide. With its high-efficiency disinfecting properties, it can eliminate a wide range of pathogens including bacteria, viruses, and protozoa. This makes chlorine the cornerstone of municipal water systems and swimming pool maintenance.
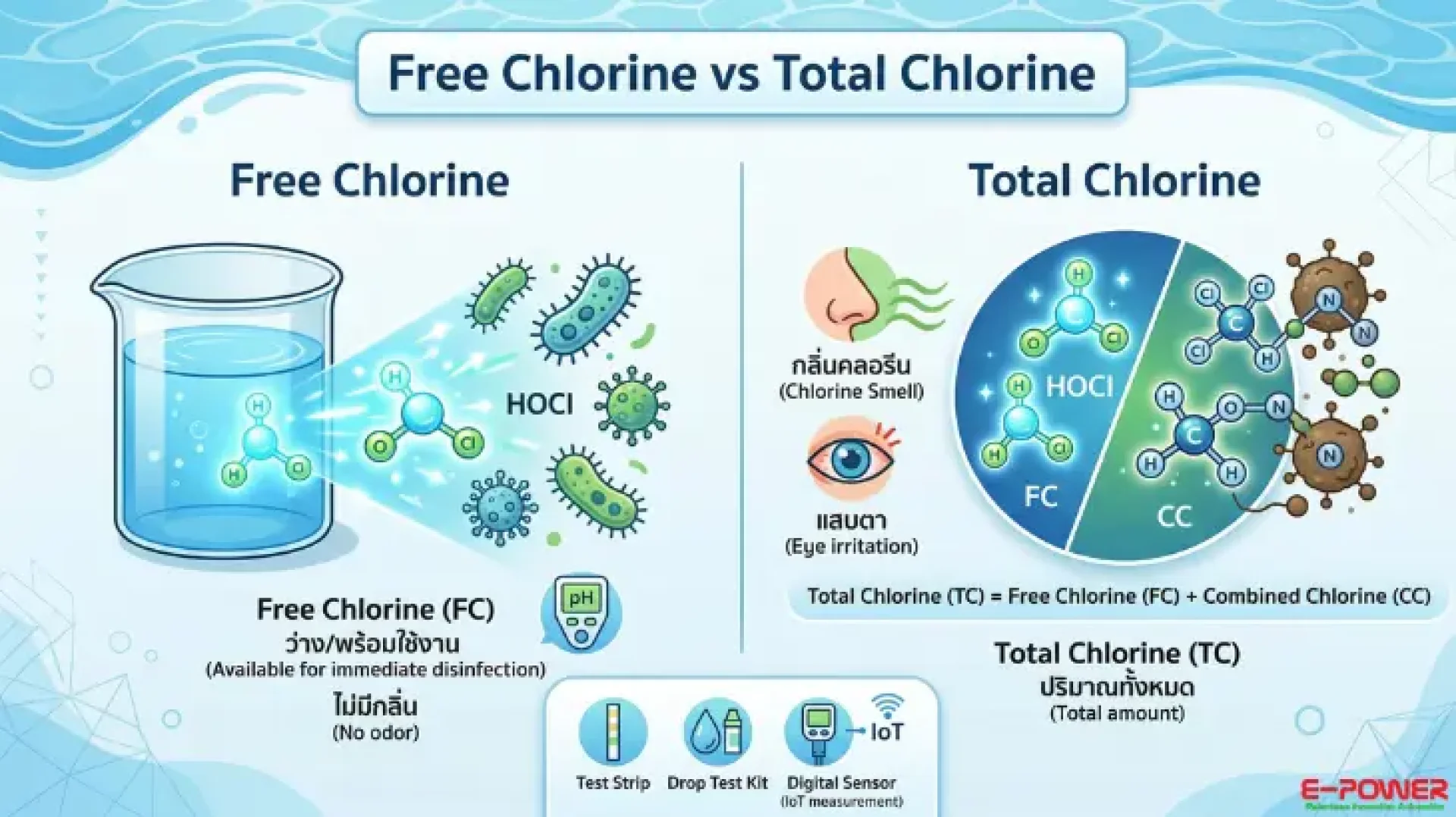
When chlorine is added to water, it undergoes hydrolysis to form Hypochlorous Acid (HOCl) and Hypochlorite Ion (OCl⁻), which are the active disinfecting agents. Hypochlorous acid is most effective under optimal pH conditions.
For precise water quality control, we must distinguish between two primary forms of chlorine:
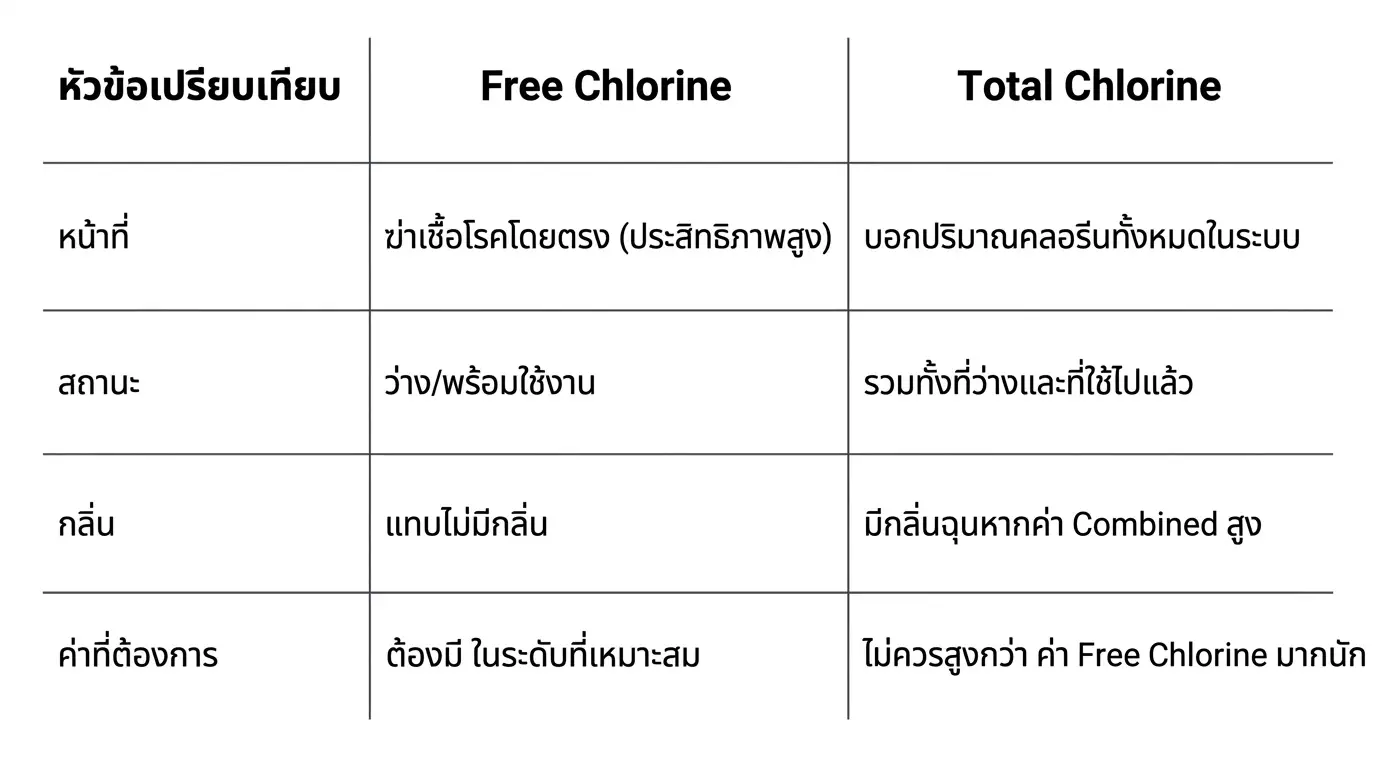
- Free Chlorine: Chlorine that is "unbound" and available for immediate disinfection.
- Total Chlorine: The sum of Free Chlorine and chlorine that has already reacted with contaminants (Combined Chlorine).
Benefits of Chlorine
Beyond water treatment, chlorine plays a vital role in various other industries:
- Cleaning Products: Used as a primary ingredient in disinfectants and detergents.
- Manufacturing Industry: A precursor in the production of paints, paper, textiles, and pharmaceuticals.
- PVC Production: Over 50% of global chlorine production is used to manufacture PVC plastic for construction and plumbing.
- Long-term Disinfection: When chlorine reacts with nitrogen compounds, it forms Chloramines, which provide longer-lasting residual disinfection in large distribution pipe networks.
Understanding the 3 Types of Chlorine
Effective disinfection is not just about the amount of chlorine added, but understanding its "form." The basic formula is:
Free Chlorine + Combined Chlorine = Total Chlorine
1. Free Chlorine
This is the amount of chlorine "ready to use" for neutralizing bacteria and viruses. It is the most reactive and effective form.
- Recommended Value: For swimming pools, it should be maintained between 2–4 PPM for effective microbial control.
2. Combined Chlorine
Formed when free chlorine binds with ammonia or organic substances, resulting in compounds called Chloramines.
- Key Characteristics: The source of the strong "chlorine smell" and eye irritation in pools.
- Ideal Value: Should be less than 0.5 PPM. If higher, it indicates the water is becoming contaminated and requires "Shock" treatment.
3. Total Chlorine
The total concentration of both available (free) and spent (combined) chlorine. If Total Chlorine is significantly higher than Free Chlorine, it indicates a high accumulation of chloramines in the water.
Summary: Free Chlorine vs Total Chlorine
Chlorine Testing Methods
Regular monitoring is crucial, especially for outdoor pools exposed to UV light and rain. There are 3 main methods:
- Test Strips: Low cost and easy to use, but lower accuracy. Best for residential use.
- Drop Test Kits: Uses chemical reagents for color comparison. Moderate accuracy.
- Digital Chlorine Sensors: Provides real-time data with laboratory-grade precision. No manual color matching required.
Water Quality Monitoring Solutions by Renke
E-Power Service Co., Ltd. offers professional-grade sensor technology from Renke designed for rigorous monitoring:
- Free Chlorine Sensor : Digital, accurate, and durable measurement of free chlorine.
- pH Sensor : Used alongside chlorine sensors to ensure chlorine performs at its peak (Chlorine works best at specific pH levels).
- Integrated System : We provide IoT data transmission systems for 24/7 monitoring via mobile devices.
Interested in installing an automatic chlorine monitoring system?
- Website: www.epower.co.th
- Email: info@epower.co.th
- LINE: @epower


